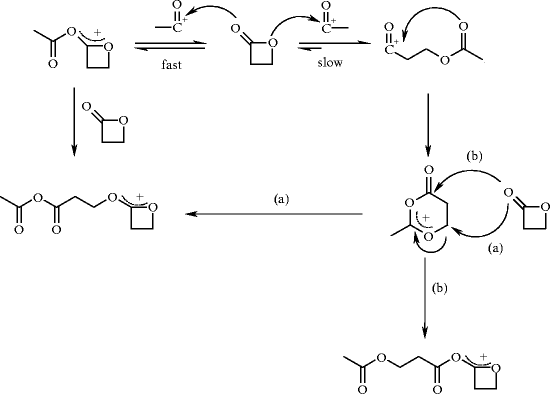
Structural Basis of the γ-Lactone-Ring Formation in Ascorbic Acid Biosynthesis by the Senescence Marker Protein-30/Gluconolactonase | PLOS ONE

Organocatalysis applied to the ring‐opening polymerization of β‐lactones: A brief overview - Khalil - 2019 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library

the Sugar and Lactone Ring Structure-Activity Relationships for the Hypertensinogenic Activity of Ouabain : Role of | Semantic Scholar
Structural Basis of the γ-Lactone-Ring Formation in Ascorbic Acid Biosynthesis by the Senescence Marker Protein-30/Gluconolactonase | PLOS ONE
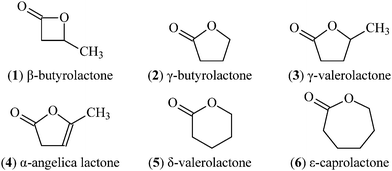
Degradable polymers from ring-opening polymerization of α-angelica lactone , a five-membered unsaturated lactone - Polymer Chemistry (RSC Publishing) DOI:10.1039/C1PY00067E

Controlled Random Terpolymerization of β-Propiolactone, Epoxides, and CO2 via Regioselective Lactone Ring Opening | CCS Chem

Molecules | Free Full-Text | Lactones 41. Synthesis and Microbial Hydroxylation of Unsaturated Terpenoid Lactones with p-Menthane Ring Systems

SciELO - Brasil - Synthesis of medium ring and macrocyclic acetylenic lactones by the ring expansion of oxabicycloalkenones Synthesis of medium ring and macrocyclic acetylenic lactones by the ring expansion of oxabicycloalkenones
Biological stains; a handbook on the nature and uses of the dyes employed in the biological laboratory. Stains and staining (Microscopy); Stains and Staining; Dyes. 184 Biological Stains 3. FLUORAN DERIVATIVES